
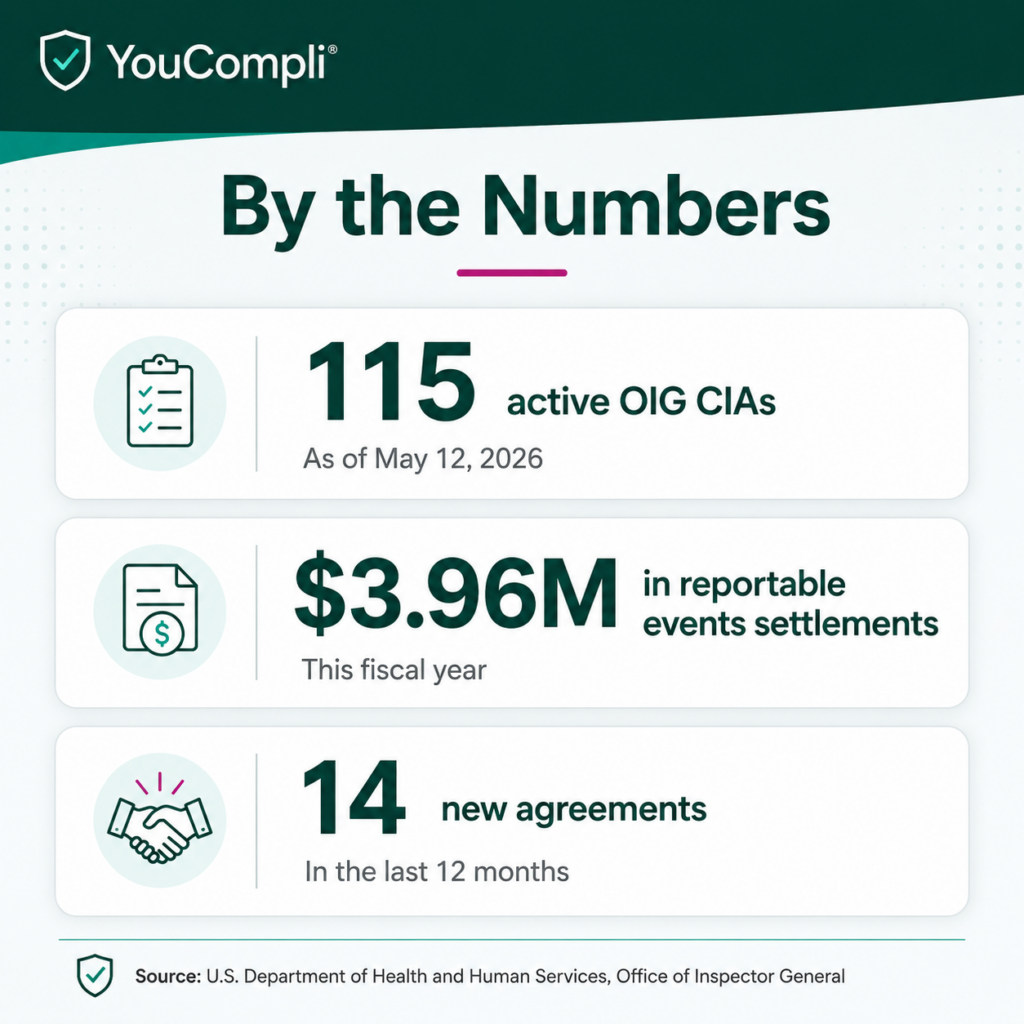
With the release of the CIA changes within the General Compliance Program Guidance (GCPG), the OIG has officially moved the goal posts. This isn’t just a new set of rules for organizations under investigation.
The May 2026 update to the Corporate Integrity Agreement (CIA) framework signals a definitive shift from passive reporting to active governance. Compliance is no longer an administrative sub-function. It’s now a board-level strategic imperative.
The OIG’s Modernized CIA: Your New Compliance North Star
Whether you’re a leader in an acute care organization, or in a non-acute care market like home health, ASCs, labs, or behavioral health, these updates represent your new “North Star.”
This blog offers a roadmap to navigating the four pillars of this modernized framework.
Pillar 1: The Board’s Mandatory Compliance Partner
What does the OIG’s mandatory independent board compliance expert requirement mean for healthcare leadership?
For years, boards could fulfill their oversight duties by simply reviewing a quarterly slide deck. Those days are over. The OIG now requires the appointment of an independent compliance expert with federal healthcare program experience to review program effectiveness and report findings directly to the board.
An expert is only as effective as the data they have to work with. To be truly “audit-ready,” your organization needs a living, centralized record of every regulatory change and the specific actions taken in response. Without this “evidence engine,” an independent expert will spend hundreds of billable hours just trying to find the paper trail. That could create a massive financial liability for the board.

Boards are also now mandated to formally respond to the expert’s findings in their annual reports. You can’t “file and forget” an audit anymore. The OIG is looking for a “culture of inquiry” where board members aren’t just receiving information but actively challenging it.
Pillar 2: Catching Every Whisper in the “Any Modality” Net
How has the OIG expanded the definition of a healthcare disclosure program beyond the traditional hotline?
The OIG has finally caught up with how the modern healthcare workplace actually communicates. A “disclosure” is no longer defined by the phone number it was called into. Now it’s defined as any report made through any modality.
The reality is, in a busy lab or ASC, a compliance concern rarely starts with a formal hotline call. It starts with an email to a supervisor, a quick Teams message between nurses, or an “off-the-cuff” comment in a hallway or breakroom.
The Roadmap Action: If a billing concern is raised in a Microsoft Teams thread but never makes it into your formal compliance log, you are in violation of the new standards.
- Modality Audit: You must ensure your intake log captures:
- Digital Channels: Email, Slack, Microsoft Teams, Zoom chat, and other messaging platforms.
- Interpersonal Channels: In-person “walk-ins” and verbal reports made to management or HR.
- The Log Requirement: Every report, regardless of how it is received, must be tracked with its nature, status, and corrective action taken.
Pillar 3: Technological Accountability and the IT Governance Mandate
What are the specific OIG reporting requirements for Generative AI and Information Technology in the 2026 CIA framework?
In perhaps the most significant update to the compliance landscape in decades, the OIG is mandating technological accountability. IT has gone from support service to a governance partner.
Set a Place for the Mandatory IT Seat at the Table
The modernized CIA framework requires that the Compliance Committee formally include IT expertise. The OIG recognizes that in a digital-first environment, governance is impossible without an understanding of the underlying infrastructure, data flows, and automated systems.
The Roadmap Action: Organizations should update their Compliance Committee Charters to include a CTO, CISO, or equivalent technical lead. This individual is responsible for bridging the gap between regulatory mandates and digital execution.

Defining and Inventorying Generative AI as a Requirement
The OIG has introduced a formal, rigorous definition of Generative AI for reporting purposes. The new definition focuses on foundation models and systems that “generate new content or inferred outputs” used in clinical, billing, or administrative decision-making.
Your Generative AI Inventory must track:
- Life Cycle Use Cases: Identifying where AI is deployed, such as automated utilization reviews, draft patient communications, clinical note summarization, or coding/billing reconciliation.
- Deployment Status: Reporting whether a tool is in the “Pilot,” “Pre-deployment,” or “Production” phase.
- Data Provenance and Security: Documenting how Protected Health Information (PHI) is shielded from being used as training data for public or foundation models.
The Risk of “Shadow AI”
The OIG is particularly focused on “Shadow AI”, meaning automated tools implemented by individual departments without the Compliance Committee’s knowledge. Under the new reporting mandates, a marketing team using an AI bot to answer patient queries or a billing department using a third-party “coding assistant” without formal approval is now a reportable risk.
By mandating these inventories, the OIG is forcing a shift from “trust” to “verification.” Compliance committees must document their review and approval of any AI tool before it impacts federal healthcare program billing or patient care. This creates a mandatory “accountability trail” that proves the organization is managing AI risks, such as algorithmic bias and “hallucinated” clinical data.

Pillar 4: The Elevated (and Independent) Compliance Officer
How is the OIG changing the independence and stature of the Compliance Officer role?
The OIG is giving the Compliance Officer (CO) role real “teeth.” The new framework explicitly states that the CO must report directly to the CEO and cannot be subordinate to the General Counsel or the CFO.
This change is about removing conflicts of interest. Legal and Finance have their own mandates, and Compliance must be free to evaluate them both.
The Roadmap Action:
- Direct Board Access: The CO must have direct, unfiltered access to the board.
- Executive Sessions: Mandatory quarterly meetings with the board, including at least one portion in “executive session” without other senior management present.
- Board Supervision: The board is now responsible for the supervision and performance evaluation of the CO, further decoupling the role from the CEO’s sole influence.
Conclusion: Your Proactive CIA Readiness Checklist: Four Tactical Steps
The OIG isn’t about making the job harder. They’re making it more technical and more transparent. To align with these modernized expectations before a regulatory knock at the door, prioritize these four tactical steps:
- Appoint an IT Lead to Compliance: Update your charter to include a technical expert who can oversee your AI and data governance.
- Conduct an AI Audit: Identify where automated tools are being used. Document what tools are being used, for what purpose, and how the data is being protected.
- Centralize Your “Any Modality” Intake: Move away from fragmented email folders. You need a single source of truth that can export a defensible log of every hallway conversation and digital message.
- Adopt the Gold Standard Early: Treat these new CIA mandates as a “Pre-Settlement Readiness Review.” Adopting these standards now proves to the OIG that your organization is committed to a culture of integrity, not just the appearance of one.
Need some help putting together your “evidence engine” and a proactive CIA strategy? YouCompli takes acute and non-acute care organizations from chaos to compliance confidence. Reach out with your questions and challenges.
Blog Glossary
- General Compliance Program Guidance (GCPG): The OIG’s definitive framework for healthcare compliance.
- Active Governance: A model where leadership and the board are directly involved in compliance effectiveness, moving beyond “check-the-box” reporting to formal responses and resource allocation.
- Corporate Integrity Agreements (CIA): Binding five-year contract between healthcare entity and the HHS Office of Inspector General (OIG), imposed as part of a civil settlement to resolve allegations of fraud or abuse.
- Shadow AI: The unauthorized or undocumented use of artificial intelligence tools within an organization without the review or approval of the Compliance Committee or IT leadership.
- Generative AI: A category of technology utilizing foundation models to generate new content or inferred outputs used in clinical, administrative, or billing decision-making.
Six More Resources
Four Ways to Engage Leaders in a Culture of Compliance – Building positive, proactive relationships with leaders is crucial to a culture of compliance. Learn four approaches to engage leaders.
10 Tips for Building a Compliance Culture in Healthcare – The tips explore how you can convince leaders that a strong compliance culture adds value to the organization.
What to Look for in Regulatory Change Management Software for Healthcare Compliance – A modern regulatory change management (RCM) system does more than just notify you when regulatory changes occur. Learn what’s really possible today and why it matters.
Your Board Committee Is Only as Effective as You Make It – Ten ways to engage your healthcare compliance board committee.
How to Use AI for Systems Thinking in Compliance Processes – Artificial intelligence is beginning to reshape the compliance landscape, but the field is still in the early stages of adoption.
Experts Weigh In: The Oversight Role of a Healthcare Board of Directors – Whether new to the field or a seasoned professional, get ideas on how support your board’s oversight responsibility for the compliance program.
About the Author
Jerry Shafran is the founder and CEO of YouCompli with years of experience in the healthcare compliance space.
He is a serial entrepreneur who builds on a solid foundation of information technology and network solutions. Jerry launches, manages and sells software and content solutions that simplify complex work. His innovations help compliance professionals focus on their core priorities.

AI Handbook for Compliance Pro’s
Video resource hub


